Stem Cell Therapy for Corneal Damage: A Breakthrough Procedure
Stem cell therapy for corneal damage represents a groundbreaking advancement in ocular medicine, specifically in addressing injuries that previously left patients with permanent vision impairment. This innovative approach leverages cultivated autologous limbal epithelial cells (CALEC) to restore the corneal surface, offering new hope for those suffering from conditions like chemical burns and infections. In a recent clinical trial led by experts at Mass Eye and Ear, stem cell treatment demonstrated a remarkable 90% effectiveness rate in recovering damaged corneas, showcasing its potential to revolutionize corneal restoration. As the procedure continues to progress, the role of ocular stem cell therapy becomes increasingly vital in treating debilitating eye injuries. With promising results from early trials, there is optimism that this method could eventually become a mainstream treatment option, lessening the burden of vision loss for many individuals.
The field of eye stem cell treatment is rapidly evolving, particularly with the introduction of new therapies aimed at repairing corneal damage. Innovative techniques, including the transplantation of limbal epithelial cells, are paving the way for effective solutions to address serious ocular injuries that have long been challenging to manage. By harnessing the regenerative properties of stem cells, medical professionals can potentially reverse conditions such as limbal stem cell deficiency, thereby restoring the eye’s integrity and function. This pioneering research not only promises to enhance corneal health but also expands the horizons of ocular care, making significant strides toward improved patient outcomes in vision rehabilitation. As we delve deeper into this promising area, the continued exploration of CAREC surgery and associated treatments could reshape the landscape of corneal therapies.
Understanding CALEC Surgery and Its Impact on Corneal Damage
Cultivated Autologous Limbal Epithelial Cell (CALEC) surgery represents a groundbreaking approach to treating severe corneal damage using the patient’s own stem cells. This innovative process involves harvesting limbal epithelial cells from a healthy eye, which are then cultivated in a controlled environment to create a graft that can restore the damaged cornea. This surgery was pioneered by Ula Jurkunas at Mass Eye and Ear, as part of a clinical trial that demonstrated over 90% effectiveness in restoring the corneal surface. Such advancements in ocular stem cell therapy signal a transformative movement in eye care, particularly for individuals who previously faced irreversible damage to their vision.
The significance of CALEC surgery extends beyond just restoring sight; it provides a viable treatment modality for those with limbal stem cell deficiency. Traditional methods, like corneal transplants, often fall short due to the inability to regenerate the necessary epithelial cells for a healthy corneal surface. By offering a solution that utilizes the body’s own regenerative capabilities, CALEC surgery fosters not only visual recovery but also a reduced risk of immune rejection, which has historically plagued transplant procedures.
Furthermore, the careful manufacturing and procedural protocols developed through the CALEC study underscore the scientific rigor behind this treatment. Mass Eye and Ear’s commitment to quality and safety ensures that grafts meet stringent criteria, allowing them to restore the cornea effectively. The positive outcomes observed in the clinical trial, including the restoration of vision and improvement in quality of life, highlight the potential impact of ocular stem cell treatments on public health. As ongoing research continues, there is hope that expanded trials will further validate CALEC surgery’s effectiveness and safety, leading to broader applications in clinical practice.
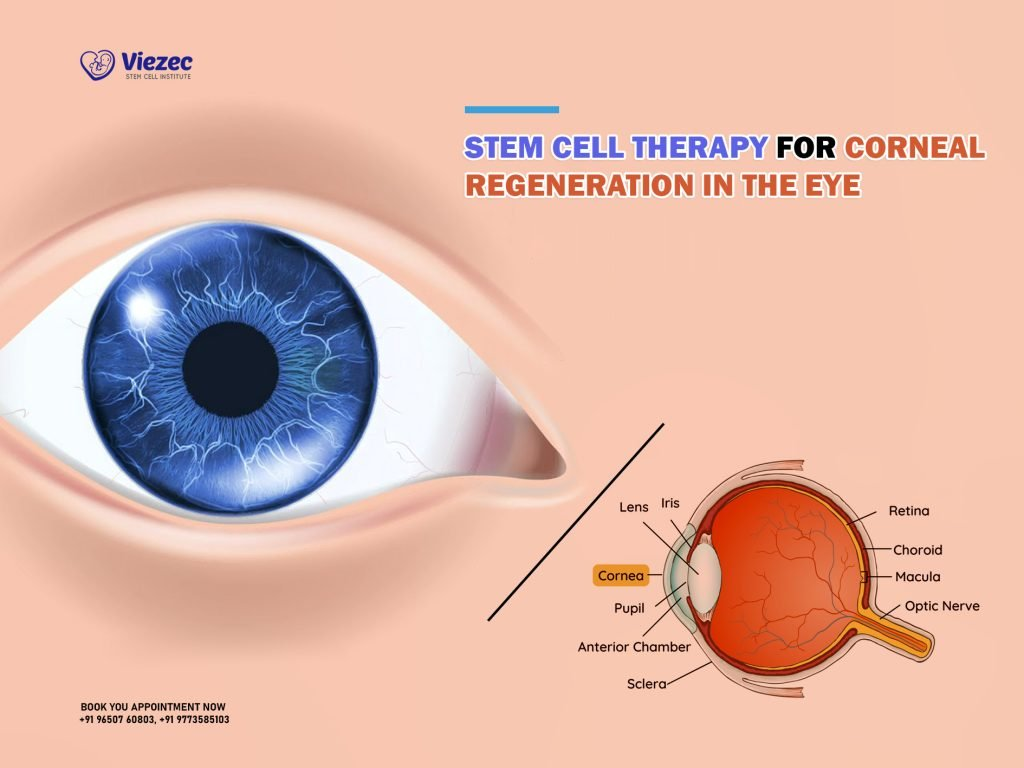
The Role of Limbal Epithelial Cells in Ocular Health
Limbal epithelial cells are crucial for maintaining a healthy corneal surface, as they are responsible for regenerating the outer layer of the cornea. These cells, located at the limbus, play a vital role in corneal homeostasis and repair following injury. When an individual suffers from chemical burns, infections, or other forms of trauma, the depletion of these cells can lead to limbal stem cell deficiency, resulting in a damaged surface that is unresponsive to conventional treatments. This condition often leads to persistent pain and significant visual impairment, emphasizing the necessity of innovative therapies such as CALEC surgery to restore corneal integrity.
With the innovation of ocular stem cell therapy, the focus is on harnessing limbal epithelial cells to regenerate a healthy corneal surface. The environment surrounding these cells is essential; they require optimal conditions for proper function and repair. Therefore, understanding the intrinsic properties and interactions of limbal stem cells within the ocular microenvironment is fundamental for advancing treatment modalities. Enhanced techniques in cell culture and grafting not only address the immediate issues of corneal damage but also pave the way for future applications in eye stem cell treatment, which could benefit a broader patient population.
In addition, the significance of limbal epithelial cells extends to the potential for allogeneic grafts in ocular treatments. Researchers are exploring the use of limbal stem cells harvested from cadaveric donor eyes, which could revolutionize accessibility to stem cell therapies for patients suffering from bilateral corneal injuries. This potential increase in donor availability would significantly improve treatment options for numerous patients who currently have limited access to stem cell therapy. The ongoing research into this area is promising, as it strives to mitigate the constraints faced by individuals with severe cornea damage.
Clinical Outcomes of Stem Cell Therapy for Eye Damage
The clinical outcomes of stem cell therapy, particularly through the CALEC procedure, have demonstrated remarkable efficacy in repairing corneal damage. In a study involving 14 patients, significant improvements were observed over an 18-month period, highlighting a 90% effectiveness in restoring corneal surfaces. Notably, these improvements were not just limited to structural integrity; many patients reported enhanced visual acuity and a reduction in symptoms associated with corneal damage. This trial, a first of its kind, establishes a new benchmark in the application of ocular stem cell therapies, providing new hope for patients who previously faced the bleak prospect of irreversible vision loss.
Moreover, tracking the progress of participants revealed that half achieved complete restoration of their corneal surface within three months, with continued improvements noted at 12 and 18 months. These outcomes indicate not only the short-term benefits of CALEC surgery but also promising long-term potential. Such developments bring forth exciting possibilities for advancing care in ophthalmology, contributing to a paradigm shift in how ocular injuries are treated and managed.
Safety profiles also highlight the potential of CALEC therapy to provide effective treatment with minimal risks. The trial reported only one adverse event—a bacterial infection in a participant, which was linked to chronic contact lens use rather than the graft itself. Overall, the low incidence of serious complications accentuates CALEC’s role as a safe and viable treatment option. As researchers continue to analyze and refine this approach, further studies will expand the understanding of stem cell therapy’s efficacy and safety, ensuring that the therapeutic benefits are accessible to a broader population of individuals suffering from corneal damage.
Future Directions in Ocular Stem Cell Research
The advancement of ocular stem cell therapy holds tremendous promise for the future of ophthalmology. Researchers are looking to broaden the applications of CALEC surgery beyond individuals with unilateral eye damage by exploring the use of allogeneic grafts derived from cadaveric sources. This shift could potentially help those patients suffering from bilateral corneal injuries, who currently have very limited options for restoration of vision. Breakthroughs in the manufacturing of limbal epithelial cells at facilities like the Connell and O’Reilly Families Cell Manipulation Core Facility could pave the way for more standardized processes that can efficiently provide the necessary grafts.
Furthermore, there is tremendous potential for collaborative research to drive innovations in ocular health. Clinical trials with larger patient populations, longer follow-up periods, and controlled randomized designs will provide deeper insights into the effectiveness of stem cell therapies in various ocular conditions. Innovations in gene editing and cell engineering could further enhance the potential of stem cell treatments, providing targeted solutions for complex eye diseases. The integration of cutting-edge technologies in ocular stem cell research represents a hopeful frontier that could drastically improve patient outcomes and quality of life.
Challenges and Considerations in Stem Cell Therapy
While the advancements in CALEC surgery and stem cell therapy bring hope, significant challenges remain. One major consideration is the complexity involved in obtaining limbal stem cells for grafting, especially in patients with bilateral damage. The requirement for a healthy donor eye complicates the process, necessitating meticulous patient selection and management strategies. Furthermore, ensuring the long-term effectiveness and safety of the grafts is paramount, necessitating continued monitoring and follow-up to identify any potential adverse effects or complications that may arise post-surgery.
Moreover, the regulatory environment surrounding stem cell therapies in the U.S. can present hurdles to the dissemination of novel treatments like CALEC surgery. Gaining FDA approval requires extensive clinical data that guarantees not just efficacy but also safety in long-term applications. Navigating this approval process will be crucial for bringing advanced ocular therapies to market, as well as for ensuring that patients have access to cutting-edge treatments. As researchers, clinicians, and regulatory bodies work together to address these challenges, the promise of stem cell therapy in restoring sight will move closer to reality.
The Importance of Multidisciplinary Collaboration in Eye Care
The successful development and application of CALEC surgery underscore the significance of multidisciplinary collaboration in delivering effective eye care solutions. The synergy between ophthalmologists, researchers, regulatory agencies, and manufacturing facilities has played a crucial role in advancing clinical trials and ensuring the high quality of grafts. By fostering an environment of collaboration, experts from various specialties can share insights, resources, and expertise to tackle the complexities associated with ocular stem cell therapy. Such teamwork is crucial in translating lab-based research into tangible clinical applications that can benefit patients.
Furthermore, engaging in partnerships with institutions like Dana-Farber Cancer Institute and Boston Children’s Hospital expands the research potential and fosters innovative approaches to ocular treatment. Collaborative research efforts not only accelerate discovery but also enhance the understanding of complex eye conditions that can benefit from regenerative therapies. As the field of ocular stem cell treatment continues to evolve, collaboration among diverse professionals will be instrumental in bridging gaps, optimizing patient care, and ultimately leading to improved clinical outcomes for individuals suffering from corneal damage.
Patient Perspectives on Stem Cell Therapy and Corneal Restoration
Understanding patient experiences and perspectives regarding stem cell therapy for corneal damage is vital in shaping treatment approaches and enhancing the overall patient care process. For many patients, the potential for restored vision through CALEC surgery represents a beacon of hope, particularly for those who have endured long-term visual impairment due to corneal injuries. Gathering qualitative data on their experiences can provide critical insights into the impact of the therapy on their quality of life and visual function, ultimately guiding clinicians in tailoring care to meet patient needs.
Additionally, empowering patients with knowledge about their treatment options is essential in fostering informed decision-making. The complexities associated with stem cell therapy, including the preparatory biopsy for the healthy eye and the subsequent surgical procedures, can evoke anxiety in patients. Clear communication regarding the benefits, risks, and expected outcomes of CALEC surgery is necessary to establish trust and improve adherence to treatment protocols. As the field progresses, prioritizing patient perspectives will play a crucial role in the successful integration of stem cell therapies into routine ophthalmic care.
Regulatory Considerations for Ocular Stem Cell Treatments
As advancements in stem cell therapy continue at a rapid pace, regulatory considerations are becoming a focal point in the successful application of these treatments. The complexities of ensuring patient safety and treatment efficacy necessitate a robust framework for evaluating novel therapies like CALEC surgery. Regulatory agencies, such as the FDA, require extensive data from clinical trials to substantiate claims of safety and effectiveness. Navigating this regulatory landscape is crucial for the successful translation of ocular stem cell therapies from research to clinical practice, ensuring that patients receive innovations with established safety profiles.
Moreover, as demonstrated by the CALEC study, gaining approval for stem cell therapies involves a thorough review and ongoing monitoring of patient outcomes. The need for long-term studies to assess the durability of treatment effects and the emergence of any delayed complications is paramount. Engaging with regulatory bodies from the early stages of trial design can streamline the process and facilitate pathways to approval. Ultimately, fostering relationships between researchers, clinicians, and regulatory agencies will be pivotal in ensuring that ocular stem cell treatments are both safe and accessible for a wider range of patients.
Frequently Asked Questions
What is stem cell therapy for corneal damage and how does it work?
Stem cell therapy for corneal damage involves using cultivated autologous limbal epithelial cells (CALEC), which are stem cells harvested from a healthy eye. This process includes taking a biopsy, expanding these cells in a lab, and then transplanting them onto the damaged cornea. This innovative approach aims to restore the corneal surface that would be otherwise considered untreatable.
How effective is CALEC surgery for corneal restoration?
CALEC surgery has shown remarkable effectiveness, with over 90 percent of patients experiencing restored corneal surfaces over 18 months. Initial results from a clinical trial indicate that 50 percent of participants had complete restoration at three months, increasing to 93 percent by the 12-month mark.
Are there any risks associated with ocular stem cell therapy for corneal damage?
As with any surgical procedure, there are risks. However, CALEC therapy currently has a high safety profile, with minimal adverse events reported. One participant experienced a bacterial infection related to contact lens use, while other issues were minor and resolved quickly.
Who is a candidate for eye stem cell treatment for corneal injuries?
Ideal candidates for eye stem cell treatment are individuals who have suffered corneal damage that has depleted their limbal epithelial cells, typically from chemical burns or trauma. Importantly, candidates must have at least one healthy eye from which limbal cells can be harvested.
What makes CALEC therapy different from traditional corneal transplants?
Unlike traditional corneal transplants which involve replacing the entire cornea, CALEC therapy focuses on restoring the corneal surface using the patient’s own stem cells. This method can potentially offer a solution for patients with severe limbal stem cell deficiency who are not eligible for corneal transplants.
How long does the CALEC process take from biopsy to transplant?
The entire CALEC process takes approximately two to three weeks after the initial biopsy is taken from the healthy eye. During this period, the limbal epithelial cells are expanded in a laboratory to create a cellular tissue graft ready for transplantation.
Is CALEC surgery currently available to all patients in the U.S.?
Currently, CALEC surgery remains experimental and is not widely available in the U.S. More extensive clinical trials are necessary before the treatment can be submitted for federal approval and offered at hospitals.
What future developments are anticipated for stem cell therapy for corneal damage?
Researchers hope to expand the application of stem cell therapy for corneal damage by establishing an allogeneic manufacturing process. This would enable uses of limbal stem cells from cadaveric donor eyes, potentially providing treatment for patients with damage to both eyes.
How does CALEC therapy contribute to the field of ocular stem cell therapy?
CALEC therapy represents a significant advancement in ocular stem cell therapy by providing a novel solution for corneal restoration. It not only showcases the potential of personalized stem cell treatments but also sets a precedent as the first FDA-approved human trial for stem cell therapy in the eye.
What should patients expect during recovery after CALEC surgery for corneal damage?
Post-surgery, patients should anticipate a period of monitoring and follow-up evaluations to assess the healing process. Published reports suggest visual improvements and restoration of corneal surface should be monitored closely, especially within the first 18 months post-treatment.
| Key Point | Details |
|---|---|
| CALEC Surgery | First CALEC surgery performed at Mass Eye and Ear; involves transplanting stem cells to restore cornea. |
| Treatment Process | Stem cells taken from a healthy eye, expanded in a lab over 2-3 weeks, and transplanted into the damaged eye. |
| Trial Success Rate | Over 90% effectiveness rate in restoring corneal surface after 18 months of monitoring. |
| Patient Results | 50% complete restoration at three months, increasing to 93% success at 12 months. |
| Safety Profile | High safety with few minor adverse events; one serious infection related to chronic contact lens use. |
| Future Directions | Plans to establish allogeneic manufacturing for wider application in patients with damage to both eyes. |
| Funding and Collaboration | Study funded by National Eye Institute; involved collaboration with hospitals and research centers. |
| Next Steps | Need for larger trials and further studies before treatment becomes widely available. |
Summary
Stem cell therapy for corneal damage represents a groundbreaking approach that could restore vision to individuals impacted by irreversible corneal injuries. The cultivated autologous limbal epithelial cells (CALEC) procedure has shown promising results, effectively restoring the cornea’s surface in a significant percentage of patients treated in a clinical trial. With ongoing research and future trials necessary for validation and FDA approval, CALEC presents hope for many patients with severe eye injuries where conventional treatments fall short. As the field of regenerative medicine evolves, stem cell therapy for corneal damage could radically change the landscape of ocular rehabilitation.